



Mounjaro (tirzepatide) is a prescription injectable medication primarily approved for use alongside diet and exercise to improve blood sugar control in adults with type 2 diabetes. Its active ingredient, tirzepatide, is also approved under the brand name Zepbound for chronic weight management in adults with obesity or overweight with at least one weight-related condition.
Contact UsTirzepatide, a dual GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) receptor agonist. This dual action helps lower blood sugar, slow stomach emptying, and reduce appetite.
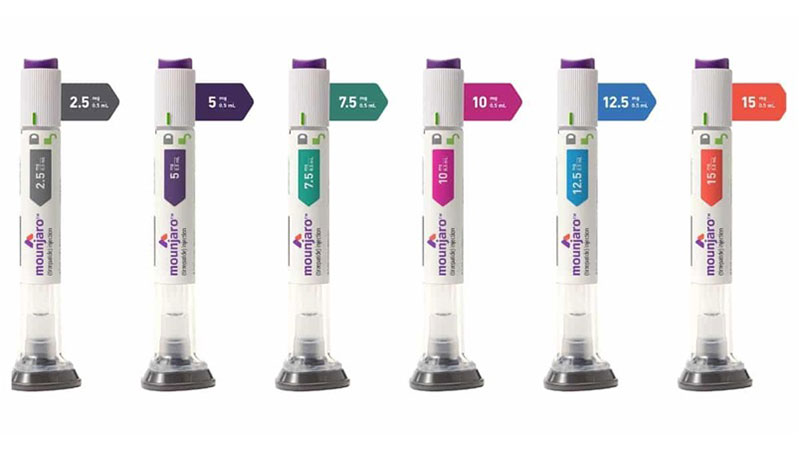
It is administered as a once-weekly subcutaneous injection in the abdomen, thigh, or upper arm. It is available in single-dose pre-filled pens or vials.
Eli Lilly and Company.
The typical starting dose is 2.5 mg once weekly for four weeks to help the body adjust to the medication and minimize side effects. The dose is then increased to 5 mg weekly, with potential further increases in 2.5 mg increments, up to a maximum dose of 15 mg once weekly, based on patient response and tolerability.
Most side effects are gastrointestinal in nature and tend to lessen over time as the body adjusts to the medication. Nausea, Diarrhea, Decreased appetite, Vomiting, Constipation, Abdominal pain, Indigestion.
Mounjaro carries a boxed warning (the FDA's most serious warning) for the risk of thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), observed in rodent studies. It is not known if it causes these tumors in humans.